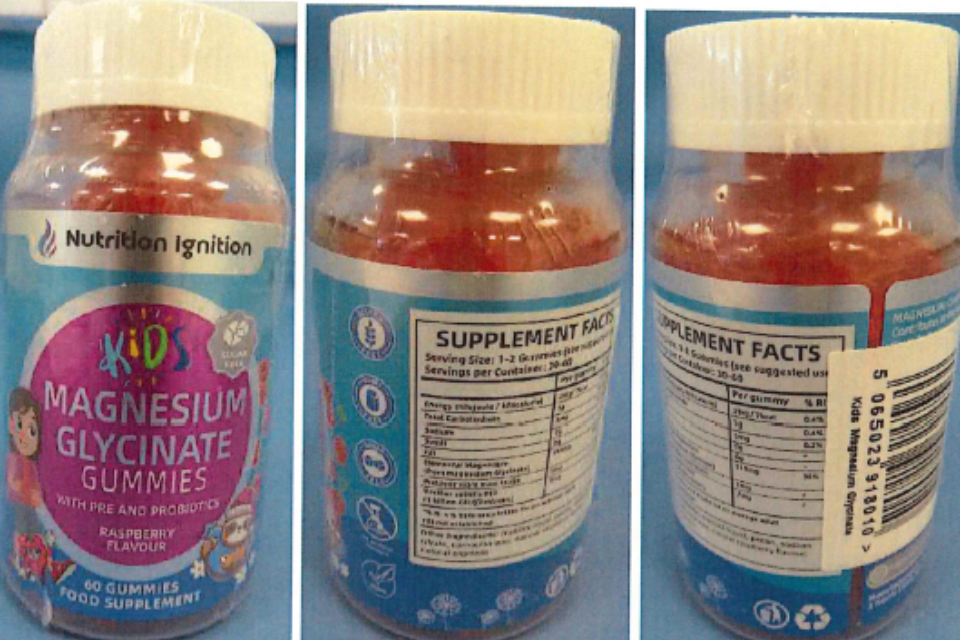
The Medicines and Healthcare products Regulatory Agency (MHRA) is advising parents and caregivers to stop all use of Nutrition Ignition Kids Magnesium Glycinate Gummies, due to the presence of undeclared melatonin, a prescription-only medicine.
Testing of two batches by the MHRA identified melatonin within the product. The gummies tested were found to contain between 1.5 and 1.7mg of melatonin in each individual gummy. Depending on the age of the child, the product packaging advised a dose of 1 or 2 gummies per day. Melatonin is not listed anywhere on the product’s packaging.
Taking too much melatonin can cause drowsiness, headache, dizziness, and nausea. Lasting harm is not expected when ingesting melatonin at high levels, and the body typically clears this within 12 hours.
This product has only been sold online as a food supplement, however, given the presence of a medicine, the MHRA took action to remove the product from sale, working with online retailers to withdraw all listings.
Anyone with this product at home is advised to store it securely in a tamper-proof container out of reach of children until they can take it to any pharmacy for disposal.
Melatonin is authorised for use in the UK as a prescription medicine for specific sleep disorders in both adults and children over the age of 6 years, where other sleep management approaches have been insufficient. If prescribed, the recommended starting dose of melatonin is 1mg for children. The dose in children can be increased to a maximum of 5mg per day before bed.
The suggested use of Nutrition Ignition Kids Magnesium Glycinate Gummies is for children aged 4 years-old and above to support ‘calm, focus, and digestion’.
Dr Alison Cave, MHRA Chief Safety Officer, said:
“We advise any parent or caregiver to stop use of this product and safely dispose of it.
“Side effects such as headache, hyperactivity, a feeling of dizziness and abdominal pain have been reported in children when melatonin is prescribed and used for its licensed indications. No serious side effects were observed in studies in children.
“Anyone who suspects that their child, or a child in their care, is having a side effect from this product is advised to stop taking it and speak to a healthcare professional and report it directly to the MHRA Yellow Card scheme.
Notes to editors
-
The detection of melatonin means this product, marketed as a food supplement, must be regulated as a medicine.
-
The MHRA cannot comment on the other ingredients listed on the label of the food supplement. The testing undertaken at MHRA labs only examined whether or not the prescription only medicine, melatonin, was present.
-
Products which fall under the definition of a medicine must hold a Marketing Authorisation (“licence”) in order to be legally sold and supplied in the UK, unless covered by a special exemption.
-
Melatonin is authorised for use in the UK as a prescription-only medicine for the short-term treatment of insomnia and jet lag in adults, insomnia in children and adolescents aged 6-17 years old with attention deficit hyperactivity disorder (ADHD) or delayed sleep wake phase disorder (DSWPD), in cases where other sleep management measures have been insufficient. Some products are approved for single use as short-term sedation under medical supervision to facilitate electroencephalograms (EEG) (a recording of brain activity) in children and adolescents aged 1-18 years old.
-
Medicines for disposal should be taken to a pharmacy. Until they can be safely disposed of, they should be stored securely in a tamper-proof container within a cupboard.
-
Suspected side effects or adverse drug reactions to any medicines should be reported to the MHRA Yellow Card scheme, either through the website (https://yellowcard.mhra.gov.uk) or by searching the Google Play or Apple App stores for MHRA Yellow Card.
-
The Medicines and Healthcare products Regulatory Agency (MHRA) is responsible for regulating all medicines and medical devices in the UK by ensuring they work and are acceptably safe. All our work is underpinned by robust and fact-based judgements to ensure that the benefits justify any risks.
-
The MHRA is an executive agency of the Department of Health and Social Care.
-
For media enquiries, please contact the [email protected], or call on 020 3080 7651.